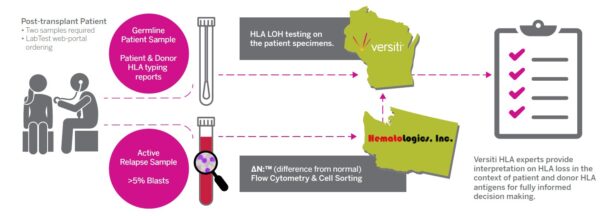
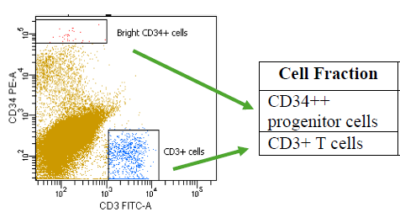
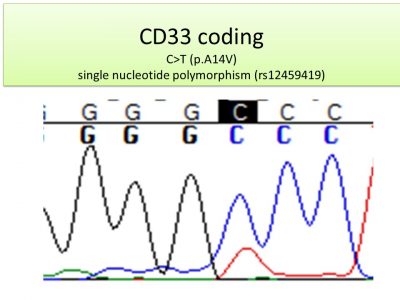
The HLA LOH assay is a multi-analytical approach leveraging the flow cytometry piece performed by Hematologics with the genomic analysis performed by Versiti. This combined approach allows the assay to differentiate sub-clonal populations with unique or unknown immunophenotypes and provides a highly sensitive assay that impacts therapeutic decisions.
Hematologics Staff 2023 Publications: Click Here
Hematologics Staff 2024 Publications: Click Here
Hematologics Staff 2025 Publications: Click Here
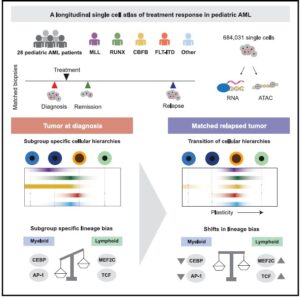
Lambo et al. use single cell sequencing analyses of clinical trial samples to identify molecular changes associated with relapsed pediatric acute myeloid leukemia. Compared to tumors at diagnosis, they find that relapsed tumors are enriched in primitive cells and typically lose myeloid transcriptional programs while adopting the transcriptional programs of other cell lineages.
Our Expertise
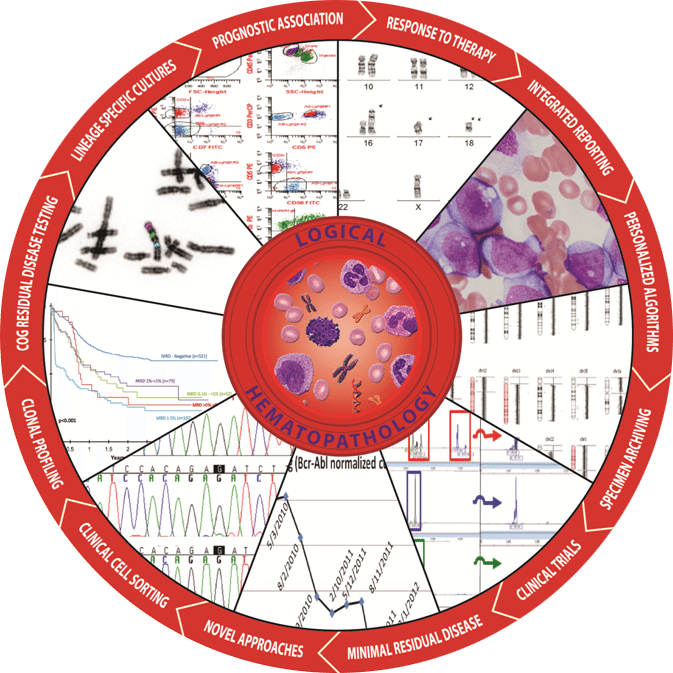
Residual Disease
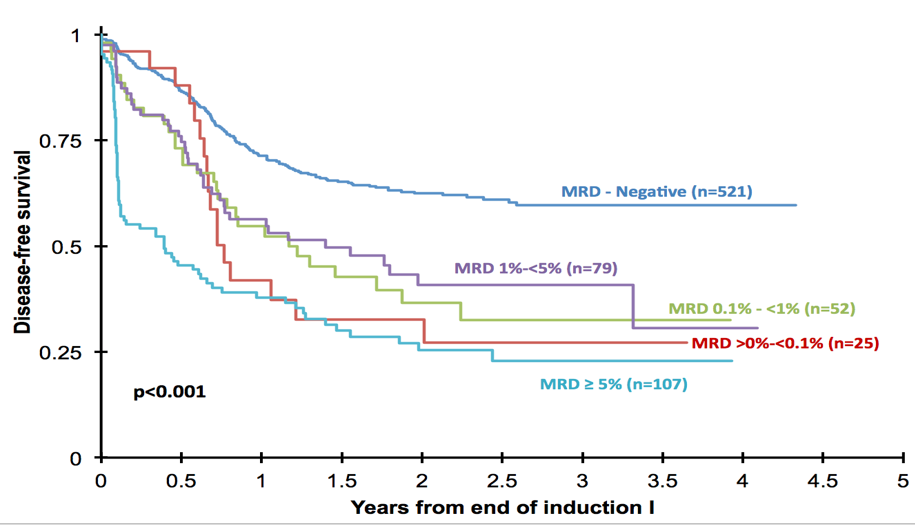
Small numbers of residual malignant cells are associated with clinical relapse at a later date. HematoLogics is the leader in residual disease testing by flow cytometry using a Difference from normal approach. We also offer a comprehensive menu of quantitative PCR assays for translocations, flow cytometric cell sorting and molecular profiling by DNA analysis.
Cytopenias /MDS
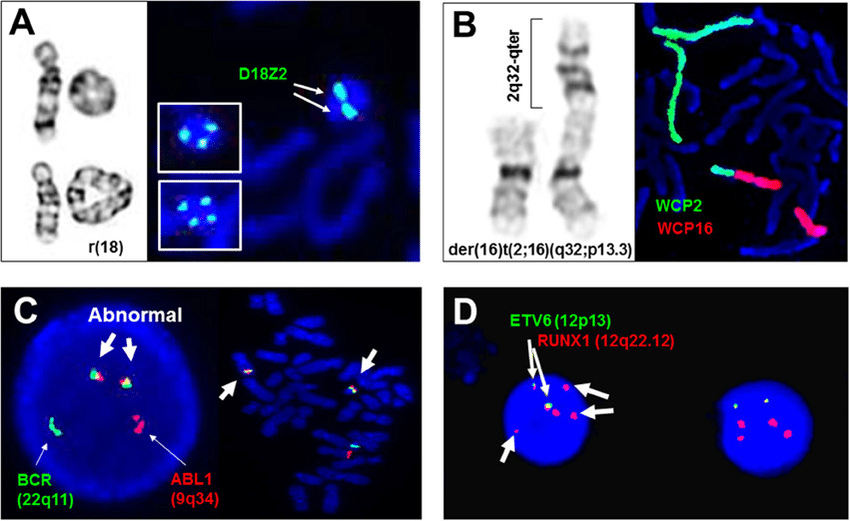
HematoLogics has found that 30% of all cytopenias are associated with abnormal myeloid cells. With our unique Flow Cytometric analysis, we use ∆N:™ (Difference from Normal) Flow Cytometry and assign an MDS Flow score that is associated with Myelodysplastic syndromes. We also offer aCGH/SNP microarray (LOH), FISH panels, cytogenetic karyotypes, comprehensive DNA mutation panels, and Next Generation Sequencing.
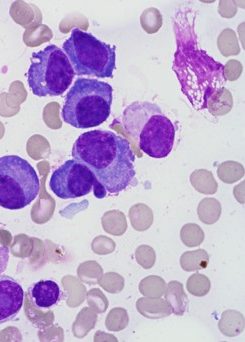
Plasma Cells
HematoLogics combines plasma cell enrichment and plasma cell sorting for FISH and aCGH/SNP microarray analysis for the diagnosis, prognosis, and monitoring of plasma cell neoplasms. For patients with concurrent monoclonal B-cell and plasma cell populations, we offer PCR IGH monoclonality profiling and MYD88 on the sorted populations to establish the diagnosis.

Biopharm
HematoLogics has extensive experience analyzing biopsies from patients on clinical trials, including those treated with novel therapies. Please contact us to find out more about how we can help clinical trial monitoring or assessment of new drug targets for the treatment of myeloid or lymphoid neoplasms.
Who We Are
HematoLogics has assembled an in-house group of experts in each diagnostic field so that all testing is performed at HematoLogics and not sent out to other specialty labs.
This speeds up analysis and permits rapid response to unexpected findings due to communication between directors on every case.
What We Do
HematoLogics provides immunophenotyping, cytogenetics, fluorescence in situ hybridization, molecular analysis studies, next-generation sequencing, and our own state-of-the-art ∆N:™ (Difference from Normal) Flow Cytometry
Send us a sample, and we will provide full interpretive reports.
Come Visit HematoLogics
Hematologics, Inc.
3161 Elliott Ave # 200
Seattle, WA 98121